iCo-007
iCo Therapeutics is developing iCo-007, a second generation antisense inhibitor targeting C-raf kinase messenger ribonucleic acid (mRNA), for the treatment of retinal neovascular diseases such as diabetic retinopathy (including diabetic macular edema). These conditions are characterized by new blood vessel growth and increased vascular permeability. Drug products that prevent the growth of new blood vessels and inhibit increased vascular permeability may have the potential to treat neovascular diseases, including diabetic retinopathy and diabetic macular edema. Read More +
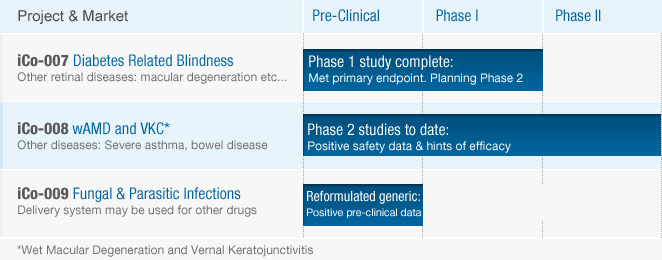
iCo-008
iCo-008 (also known as Bertilimumab or CAT-213) is a human immunoglobulin monoclonal antibody targeting eotaxin-1, a member of the chemokine family of proteins, that acts as messenger molecules between the cells of the immune system.
Eotaxin-1 is released by various cells, including eosinophils, epithelial cells, fibroblasts, endothelial cells, T-lymphocytes, monocytes and macrophages. During an allergic response the levels of eotaxin-1 are elevated. This attracts eosinophils, a type of white blood cell, into tissues where they can degranulate causing tissue damage that occurs in a variety of allergic disorders. This condition, known as eosinophilia, can occur in a number of disorders, such as allergic ocular disease of conjunctiva and cornea (conjunctivitis and keratoconjunctivitis), asthma, allergic rhinitis, atopic dermatitis, and other inflammatory disorders, such as inflammatory bowel disease and Crohn’s disease. Read More +
iCo-009
iCo Therapeutics owns the worldwide exclusive rights to iCo-009, an oral reformulation of Amphotericin B for sight and life threatening diseases. iCo-009 also represents a new drug delivery technology with the potential to reprofile other IV administered drugs to the oral route of administration.
Amphotericin B (AmpB), the gold standard for systemic antifungal drugs, is one example of a well established, highly efficacious systemic antifungal drug that has a 50 year history of intravenous therapy. AmpB formulated as a micellar dispersion (Fungizone® FZ) for IV use, remains one of the most effective agents in the treatment of systemic fungal infections, yet no oral formulations are currently commercially available. Read More +